Cancer in the UK - Digital Data Toolkit
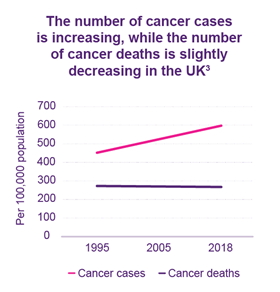
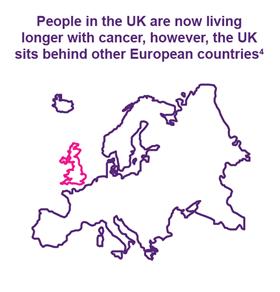
Cancer survival in the UK has improved over recent years, with more people living five years or more with the disease than ever before.1 However, despite this progress, cancer remains one of the UK’s leading causes of death.2 More can be done to improve cancer outcomes for those affected. By looking at how the UK compares to other European countries, we can begin to consider how this can be achieved.
This digital data toolkit brings together data from the Institute for Health Economics (IHE)’ Comparator Report on Cancer in Europe, to enable comparisons of the cancer outcomes achieved in the UK with that of other European countries.
In addition to the data provided by the IHE, the ABPI undertook a series of interviews with key stakeholders from across the cancer policy landscape to gain their perspectives on the data as well as their priorities for the future of cancer care in the UK. A summary of the key themes emerging from these interviews can be found in the ‘UK Stakeholder Perspectives’ page.

References:
1. NHS (2019) NHS Long Term Plan. p. 56
2. Office for National Statistics (2020) Leading causes of death, UK: 2001 to 2018.
4. Hofmarcher, T. et al. (2020) UK in Focus: Comparator Report on Cancer in Europe 2019 - Disease Burden, Costs and Access to Medicines. IHE: Lund, Sweden, slides 18 - 24

Cross-sector insights into improving outcomes for cancer patients.

This toolkit brings together data included in the Comparator Report on Cancer in Europe 2019 developed by the Swedish Institute of Health Economics.

An overview of incidence, mortality, survival, spending and uptake data for all cancers.

An overview of incidence, mortality, survival, spending and uptake data for breast cancer.

An overview of incidence, mortality, survival, spending and uptake data for prostate cancer.

An overview of incidence, mortality, survival, spending and uptake data for colorectal cancer.

An overview of incidence, mortality, survival, spending and uptake data for lung cancer.

An overview of incidence, mortality, survival, spending and uptake data for pancreatic cancer.